The GABA Receptor
This is a deep dive on arguably the most important receptor for our job as anaesthetists.
Some of this is examinable material, but mostly it's just really interesting.
The main switchboard
There are a multitude of important receptors in the world of anaesthesia, from antiemetics hammering away at serotonin receptors in the chemoreceptor trigger zone, to local anaesthetics inducing radio silence at voltage gated sodium channels.
And don't get me started on glycopyrrolate and neostigmine...
But few receptors have played such an influential role as the GABA-A chloride channel, which essentially appears to be the master switch for consciousness, amnesia and anxiolysis in the central nervous system.
Which drugs act on the GABA-A receptor?
- Propofol
- Etomidate
- Thiopentone
- Barbiturates
- Volatile anaesthetics
- Benzodiazepines
- Alcohol
- Neurosteroids
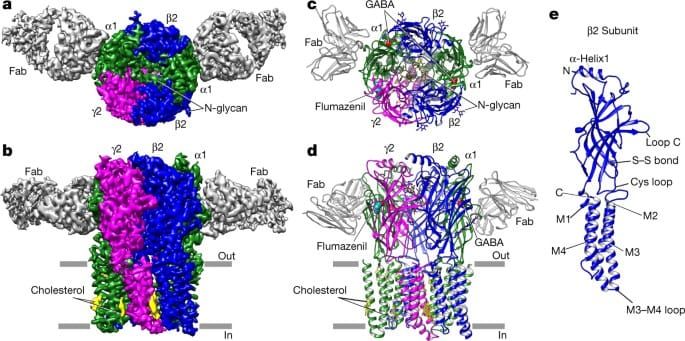
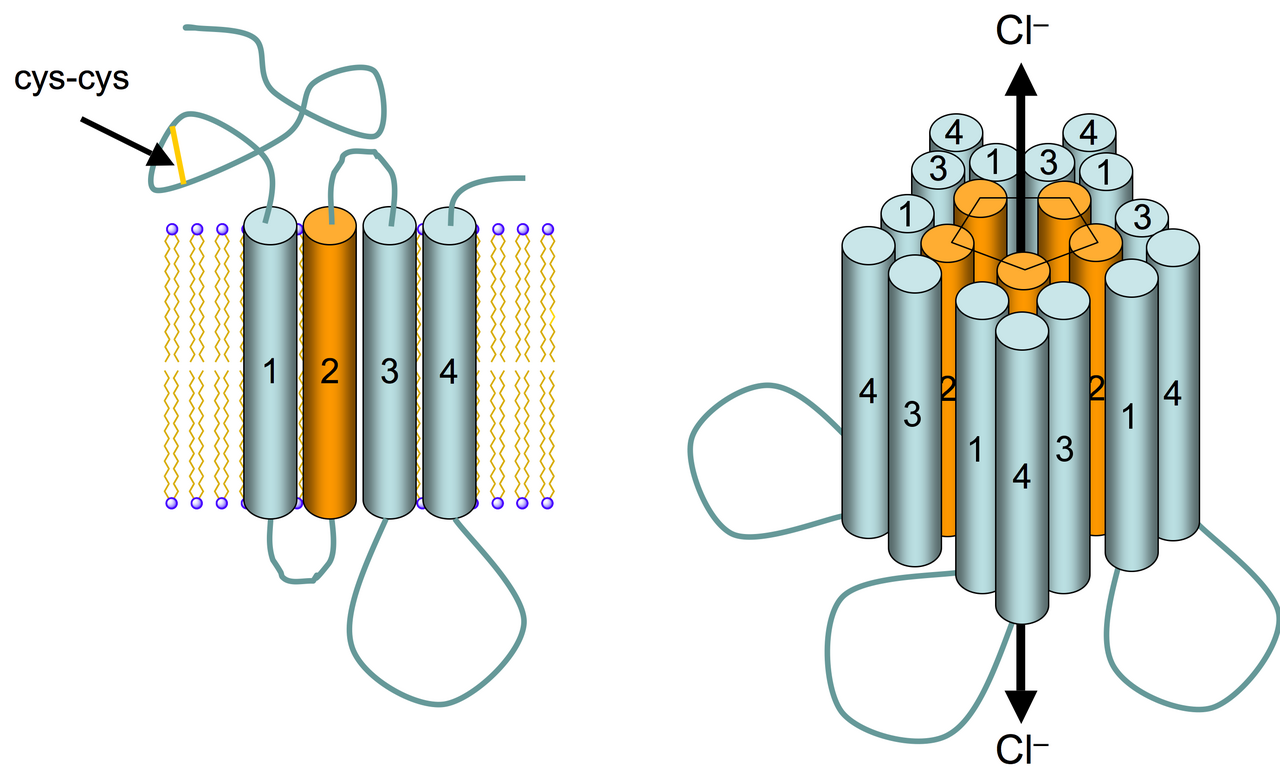
What does it look like?
The GABA-A receptor is in the same Cys-loop receptor superfamily as nicotinic acetylcholine receptors, glycine receptors and 5-HT-3 receptors.
It is:
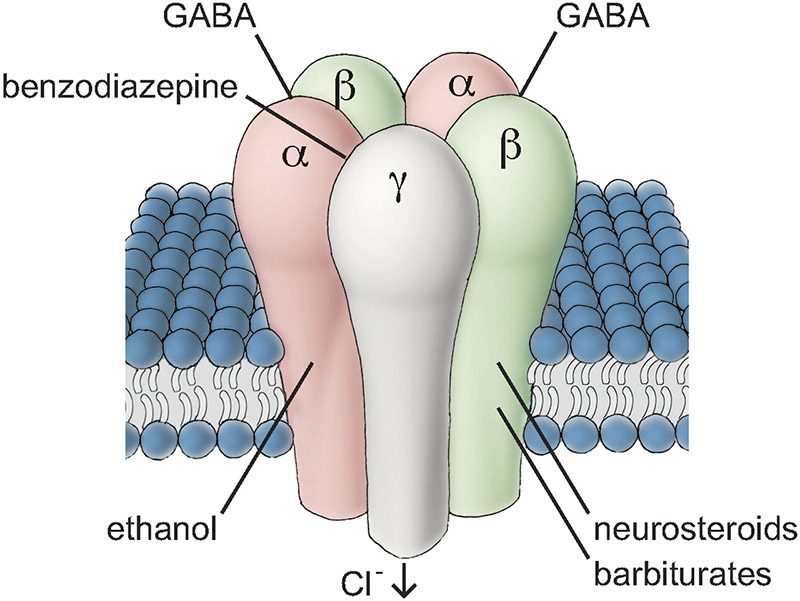
- A transmembrane pentamer of five protein subunits
- Arranged around a central hydrophilic chloride channel
The beauty of multiple subunits is the possibility of many different combinations to serve different purposes, and the most common arrangement in the adult brain is:
- 2 α subunits
- 2 β subunits
- 1 γ subunit
Specifically the arrangement around the central pore is β2, α1, β2, α1, γ2 in the most common receptor configuration.
Tell me all the different subunits
Ha. Gladly.
- α1 to 6
- β1 to 3
- γ1 to 3
- δ
- ε
- θ
- π
- ρ 1 to 3
You're welcome.
This is why different drugs that all act on the GABA-A receptor can produce wildly different clinical mixtures of:
- Hypnosis
- Amnesia
- Anxiolysis
- Sedation
- Anticonvulsant activity
Because they all tweak different receptor variants at different sites in different ways, depending on which subunits are present.
What do each of the alpha subunits do?
We think the following relationship exists:
- α1 = sedation and amnesia
- α2 = anxiolysis
- α3 = muscle relaxation
- α5 = memory and cognition
Note this is completely separate to the 'alpha' adrenergic receptors, where alpha 2 adrenoceptors also cause an element of sedation. Here we're specifically talking about the alpha subunit of the GABA-A receptor.
Don't think of the GABA-A receptor as a single receptor, it's more like a family of receptors that produce a spectrum of clinical effects depending on which subtypes are stimulated and where.
Part of the reason neonates, adults and the elderly all respond differently to anaesthetic agents and sedatives is because they express receptors with different subunits and behaviours.
How does it work?
The GABA-A receptor is the brain’s major fast inhibitory receptor.
It is a ligand-gated chloride channel.
- When GABA binds extracellularly at the interface between the β and α subunits, the receptor changes conformation
- The chloride channel opens and chloride ions flow into the cell
- This hyperpolarises the neuron
- Neuronal excitability drops*
- The volume of nervous transmission in the CNS decreases
It doesn't switch anything off, it just amplifies the inhibitory pathways that are already going on inside the central nervous system.
"Anaesthesia is a disruption of thalamocortical network connectivity rather than simple neuronal silence."
Consciousness is a network phenomenon, which needs a lot of coordination, and anaesthesia scrambles this. It is this coordinated activity that is needed to maintain awareness and create memories, hence the amnesic and sedating effects of GABA agonists.
It's a bit like turning the volume down on a radio. The radio is still on, and still receiving signals, it's just too quiet to hear.
*Super technically speaking, it actually changes the probability that neuronal networks can sustain coordinated activity.
A spot of history
The humble amino acid, gamma-aminobutyric acid was found to be present in brain tissue in the 1950s.
Initially it was thought to be an innocuous metabolic byproduct and so wasn't given much attention, however it quickly became apparent that this molecule was in fact the OG inhibitory neurotransmitter in the mammalian central nervous system.
It is produced through decarboxylation of glutamate, by the unimaginatively named glutamate decarboxylase enzyme.
And of course, as soon as something is found to be important, it becomes a supplement that you can buy for extortionate prices to support your 'neurotransmitter wellbeing'.

Fast forward to the 1970s and it became apparent that there were at least two classes of GABA receptor:
- GABA-A, a fast ionotropic ligand-gated chloride channel
- GABA-B, a slower metabotropic G protein-coupled receptor that increased potassium conductance
It was Norman Bowery's work with baclofen that was crucial to establishing GABA-B as a distinct class of receptor, with important implications in muscle relaxation and spasticity as well as epilepsy.
Then in the 1980s and 1990s it all got terribly exciting as we discovered that general anaesthesia was no longer a nonspecific membrane phenomenon (see Meyer-Overton hypothesis), but we now actually had a specific protein target that explained the effects.
Ins and outs
There's an important distinction to make in the GABA-A receptor family. There are those receptors found in the synapse (synaptic) and those outside of it (extrasynaptic).
- Synaptic receptors usually contain γ subunits, are fast-acting and cause rapid, phasic inhibition of post-synaptic action potentials
- Extrasynaptic receptors meanwhile have δ units and provide a slower, tonic inhibition
Where else are they?
GABA-A receptors are most abundant in the cerebral cortex, thalamus, hippocampus and brainstem, but they're all over the place.
Where else do you find GABA-A receptors?
- Spinal cord
- Retina
- Brainstem
- Autonomic ganglia
- Immune cells
- Airway smooth muscle
- Endocrine tissue
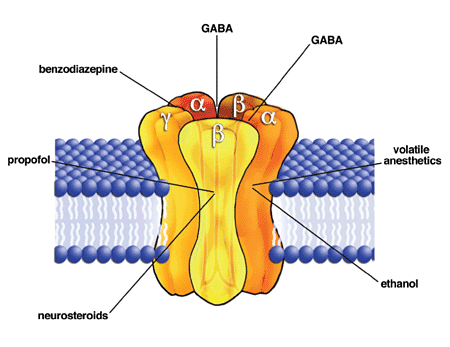
Where do our drugs bind?
So the endogenous GABA molecules bind at extracellular binding sites stuck between β and α subunits.
Most of our drugs do something different. They bind at different sites on the subunits, and adjust the frequency or duration of channel opening, when GABA binds.
This is important, because it means our drugs aren't strictly 'agonists' of the receptor, they're allosteric modulators. They still need GABA to be present to do the job of opening the channel, the drugs just amplify the effect.
Benzodiazepines
- Bind at the α/γ interface
- Increase frequency of channel opening
Barbiturates
- Bind in a variety of transmembrane regions
- Increase the duration of channel opening
- At high concentrations can directly gate the receptor open
This last point is partly why barbiturates are more dangerous, and exceptionally helpful at treating status epilepticus.
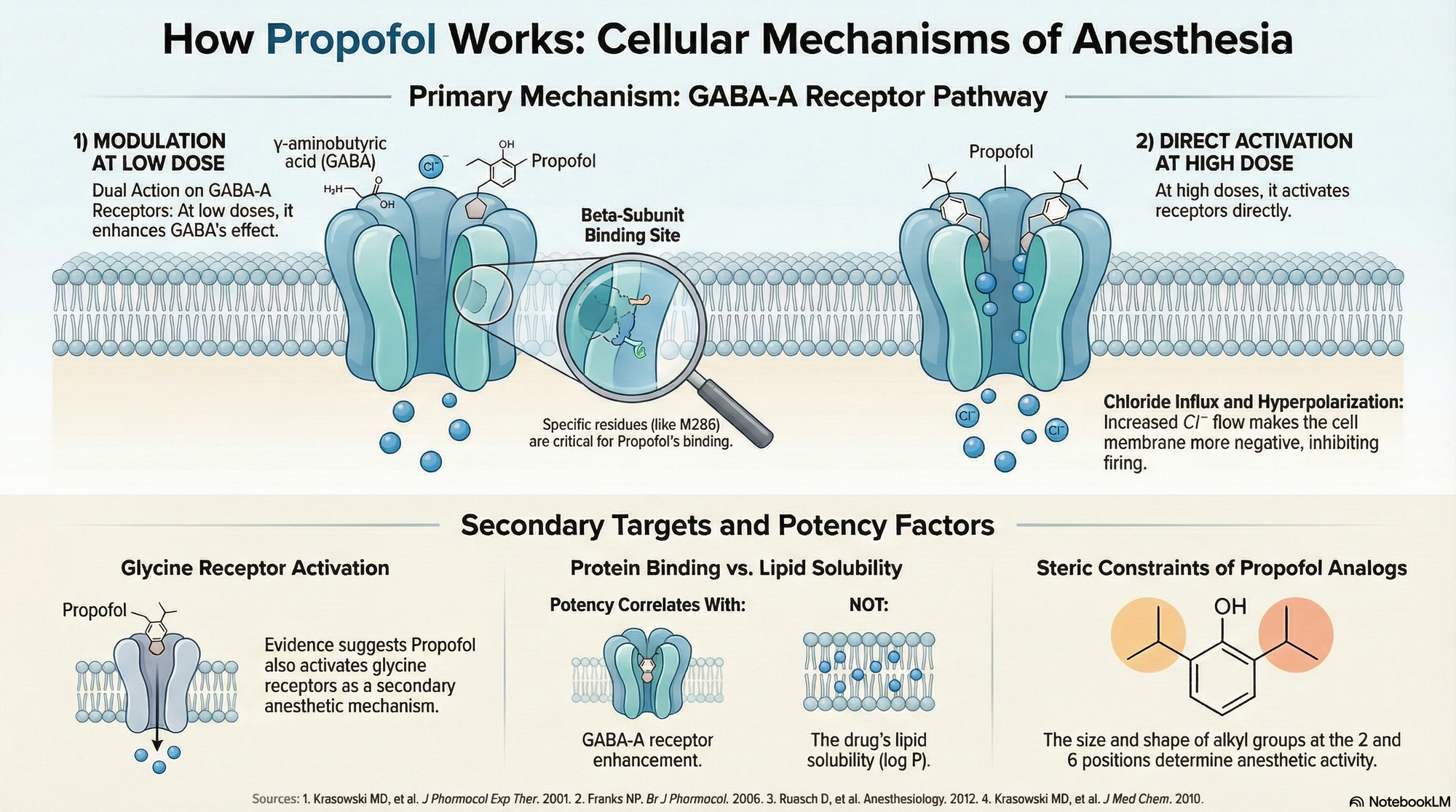
Propofol
- Binds at transmembrane β-subunit sites
- Can directly gate the channel at higher doses
- It seems that receptors containing β3 subunits are particularly useful for hypnosis and anaesthesia*
*Mice with β3 mutations are resistant to propofol anaesthesia.
See awesome image at the bottom of this post.
Etomidate
- Binds to β2 and β3 subunits
- Also causes adrenal suppression through a completely separate mechanism
Remember no drug is completely 'clean', there is almost always activity at other receptors that will cause side effects of varying clinical significance.
Just look at all the receptors ketamine hits.
Volatile agents
These are much more pharmacologically promiscuous, acting at an array of different receptors.
The current theory is that volatile agents bind directly to hydrophobic pockets within ion channel proteins.
They enhance GABA-A signalling via allosteric modulation but also affect:
- Glycine*
- Potassium
- NMDA
- A variety of others
It is the variety of action at these different receptors that is thought to be why sevoflurane, isoflurane and desflurane all have different personalities.
*Volatiles strongly enhance glycine receptor activity in the spinal cord and brainstem, suppressing spinal reflexes to a greater extent than other anaesthetic agents.
The definition of 'MAC' where patients don't flinch to a surgical incision, is largely thanks to these spinal effects of volatiles, rather than a cerebral effect of the drug.
The historical theories that anaesthetic activity was all to do with lipid-solubility now start to make sense, as most of these drugs bind at transmembrane domains on the receptor, inside the lipid bilayer, so they need to be fat soluble in order to work.
What happens with alcohol?
You've probably noticed that your patient who claims to drink 'one or two glasses' of wine each night is taking rather a long time, and several syringes, to go to sleep.
Ethanol acts partly through GABA-A receptors, but also through a host of other receptors through a messy cocktail (pun intended) of effects that we still haven't fully elucidated.
In the immediate acute phase, alcohol is a sedating agent, and will reduce the anaesthetic requirement for sedation, however chronic exposure to ethanol has the opposite effect, altering subunit composition and receptor density.
Chronic alcohol exposure leads to:
- Reduced expression of α1 subunits
- Reduced expression of δ-containing extrasynaptic receptors
- Increased expression of α4 and α6 subunits
When you lose the GABA-mediated control of activity you lose control over hypothalamic function, autonomic tone and stress control.
This is why you see the profound hypertension, sweating and tachycardia in patients violently withdrawing from alcohol.
This disruption in subunit expression is also thought to be why chronic alcohol use leads to unpredictable responses to benzodiazepines and anaesthetic agents.
Tell me about neurosteroids
Yeah this is cool.
So neurosteroids are rapid acting steroid neurotransmitters that act within seconds to minutes, rather than the hours to days that are usually associated with endogenous steroids.
They're produced in the adrenals, ovaries, testes, placenta as well as the brain itself.
Key endogenous neurosteroids include:
- allopregnanolone
- tetrahydrodeoxycorticosterone (THDOC)
- pregnenolone sulfate
The most clinically relevant for anaesthesia is allopregnanolone, but they all strongly modulate extrasynaptic GABA-A receptors.
As a result you get different responses to GABA activity when these neurosteroids are floating around, such as in pregnancy and under extreme stress.
Acute stress increases allopregnanolone production, which may act as a compensatory inhibitory mechanism.
What happens in pregnancy?
Progesterone levels rise dramatically, dragging allopregnanolone levels with it.
- This means an increase in tonic GABAergic inhibition
- We think this is why (at least in part) parturients need less general anaesthetic than their non-pregnant counterparts.
- After delivery the levels of neurosteroid drop rapidly, which can cause a withdrawal picture of anxiety, insomnia and seizure vulnerability
But that could also just be the exhaustion of having had a baby.
Other things that neurosteroids may play a role in
- ICU delirium
- Postoperative cognitive dysfunction
- Emergence phenomenon
- Catamenial epilepsy
- Postpartum depression
The key here is 'may'.
Here's an awesome image
References and Further reading

Other cool posts



The Toolkits
Everything you need to smash the exams, all in one place.


Just a quick reminder that all information posted on Anaestheasier.com is for educational purposes only aimed at trained professionals, and it does not constitute medical or clinical advice.
Anaestheasier® is a registered trademark.





