Nitrous Oxide and Entonox

Take home messages
- Nitrous is a useful analgesic and mild sedative addition to your armoury
- It's terrible for the environment, but probably not as terrible as desflurane
- Be wary of accidental hypoxia
We anaesthetists are often a rather opinionated bunch, and we all have our own views of different strengths on whether or not we like things like desflurane, the McCoy blade or trauma lists.
The same goes for nitrous oxide. Some love it, some loathe it, but we all have to know about it, so buckle up.
What is nitrous oxide and what is it used for?
Nitrous oxide is a sweet-smelling, colourless inorganic gas, discovered in the 1770s by Joseph Priestly.

It is used as an adjunct in general anaesthesia to reduce anaesthetic requirements and add analgesia. It also serves as a main form of analgesia in labour and other procedures such as joint relocation in emergency department and dental extractions.
It can also be used in dermatology as cryotherapy for removal of superficial lesions.
How does Nitrous work?
- Positive allosteric modulator of GABAa receptor
- Antagonist at NMDA receptor
- Some opiate receptor agonism may explain analgesic effect
How is it made and stored?
Nitrous is generally used as a vapour (or gas when above 36.5°C - see below) to provide its useful pharmacodynamic and physical properties.
However unlike the other commonly used medical gases such as oxygen and medical air, nitrous is stored in french blue cylinders as a liquid, with important implications on how you measure how much is inside.
What pressures are cylinders stored at for each gas? Which gas is different?
- Gas cylinders are stored at 13 700 kPa
- Nitrous is stored at 4400 kPa as a liquid with vapour phase above it
Liquid and vapour
A vapour exists when a substance is in its gaseous phase but below its critical temperature.
The critical temperature is the temperature above which a gas cannot be compressed back into a liquid, no matter how much pressure you apply.
Nitrous oxide has a critical temperature of 36.5°C.
This means that below this temperature, applying a whole load of pressure will squish it into a liquid, but above this temperature, it'll stubbornly remain as a gas.
(You might notice that this means you technically inhale nitrous as a vapour, but exhale it as a gas.)
(Zero clinical relevance but 100 nerd points.)
How is this relevant to our cylinder?
The liquid nitrous in the gas cylinder naturally will have a layer of vapour above it, as some of the liquid molecules at any given time will have enough energy to evaporate into the vapour phase.
By definition, the remaining space in the cylinder, once saturated with vapour, will sit at the saturated vapour pressure for nitrous oxide, which at 20°C is around 5200kPa, and in a cylinder at 15°C will be around 4400kPa.
As you then use the cylinder, and remove this vapour to deliver to the patient, you reduce the vapour pressure, which is then immediately topped back up by freshly evaporated liquid, and therefore the saturated vapour pressure is maintained.
You might notice the issue here.
A pressure gauge stuck onto this cylinder will display the same pressure - 4400kPa - right up until the point all the liquid is gone. Then once the liquid is gone, and as the last bit of vapour is used up, the pressure will rapidly plummet to zero. Not overly useful for measuring how much nitrous is left in the tank.
- Therefore nitrous cylinders are weighed and compared to their tare weight to determine how much liquid remains, where tare weight is simply the weight of an empty cylinder
That's why it matters.
However for the wall supply, everything is a gas.
What pressures are delivered via the wall supply?
- Gases are supplied at 400 kPa (4 bar) including nitrous
- Medical air is supplied at 700 kPa (7 bar) when used for driving surgical equipment
Manufacture
For some reason the examiners for the FRCA have decided that it's crucially important you know exactly how nitrous is manufactured.
Presumably for the inevitable transfer scenario where your ambulance breaks down and you run out of analgesia, but luckily there's a bag of ammonium nitrate by the side of the road and you've found a camp stove...
How is nitrous oxide manufactured?
- Ammonium nitrate is heated to 250°C
- NH4NO3 —> N2O + 2H2O
What are the key pharmacological properties of nitrous oxide?
Unfortunately, as with many aspects of revising for anaesthetics, this just requires some memorising of numbers. Good luck.
Boiling point
- -88.5°C
Critical Temperature
- 36.5°C
This means that above 36.5°C it cannot be compressed back into a liquid, no matter how much pressure is applied, and it is therefore a gas
Below this temperature, but above the boiling point, it can be compressed back into a liquid with sufficient pressure, and therefore it is called a vapour
- Nitrous is therefore the only vapour that is inhaled as a vapour, and exhaled as a gas
Molecular weight
- 44
This low weight means it diffuses very rapidly and extensively throughout the body
Saturated vapour pressure
- SVP varies with temperature, so the standard temperature used for comparison between gases is 20°C
- The SVP of Nitrous at 20°C is 5200kPa
- The SVP in a cylinder at 15°C is 4400kPa
- At its critical temperature the SVP is called the critical pressure
- For nitrous this is 7200kPa
Blood gas coefficient
- 0.47
- Measure of speed of onset
- Nitrous is fast in it's onset and offset, making it useful for rapid analgesia
Oil gas coefficient
- 1.4
- Measure of potency
- Nitrous is not potent at all, requiring a MAC of 103% (only possible in a hyperbaric chamber)
What are the effects of nitrous oxide on the body?
Like most gaseous agents we administer to our patients, the majority is simply exhaled unchanged, however nitrous has a number of important effects on a variety of organ systems on its journey through the body.
Cardiovascular
- Mild suppression of myocardial contraction
- Increases pulmonary vascular resistance*
- No significant drop in blood pressure as sympathetic activation maintains SVR
*Ideally avoid in pulmonary hypertension
Respiratory
- Minor reduction in tidal volume but increased respiratory rate, leaving minute ventilation the same
- Diffusion hypoxia*
*By dissolving so much faster than oxygen, it's possible to fill the alveoli with nitrous and dilute out the oxygen, generating a hypoxic gas mixture
Neurological
- Analgesia, particularly above 20-25% concentration
- Hypnosis above 70%
Metabolic
- Oxidises the cobalt ion in vitamin B12, preventing vB12’s action as a cofactor for methionine synthetase
- This leads to megaloblastic anaemia and bone marrow dysfunction
- Teratogenic
GI/GU
- Emetogenic
- Expansion of gas filled spaces - the bowel will distend over time if nitrous used in general anaesthesia, which can make it more difficult for the surgeon
- Ideally avoid in laparoscopic procedures
- Non-tocolytic*
*Can be given in general anaesthesia for emergency caesarean section to reduce the amount of volatile agent required, and therefore reduce the uterine relaxation effect seen with agents like sevoflurane
What are the advantages and disadvantages of using nitrous oxide?
Advantages
- Potent analgesic
- Weak anaesthetic
- Reduces required MAC of other volatiles
- Low blood gas coefficient means rapid onset and offset
- Increases the concentration of other agents and accelerates induction via the second gas effect
Disadvantages
- High diffusion capacity 25x that of nitrogen means it fills non compliant air filled cavities and increases pressure
- Will distend compliant air filled spaces
- Diffusion hypoxia
- Emetogenic
- Toxic
- Megaloblastic anaemia due to RNA and DNA synthesis dysfunction
- Known to be teratogenic in rats
- Neurotoxicity - causing Subacute Combined Degeneration of the Cord and Dorsal column dysfunction
What is diffusion hypoxia?
- At the end of anaesthesia, when the N2O is switched off, the large amount of N2O diffusing rapidly into the alveolus effectively dilutes the oxygen in the alveolus, and can result in a hypoxic gas mixture
- It is the opposite of the second gas effect
- If the patient is adequately preoxygenated with 90-100% oxygen this doesn’t have any clinical impact
Gas and Air
The midwife's best friend, Gas and Air or Entonox is a 50/50 mixture of nitrous and oxygen, stored in white and blue cylinders, usually with some sort of scuba gear attached for a poor labouring woman to cling to.

You might notice it has a pressure gauge on the top.
That's because entonox is a gas in a cylinder, unlike nitrous oxide, because when you make entonox by bubbling one gas through the other, you form a curious mixture that behaves differently to what you expect by just looking at each of the ingredients.
(This is called the Poynting effect)
Much like the other cylinders of gas, it is stored at 13 700 kPa and has a pressure gauge to measure its contents.
The key thing to watch out for is not to let entonox cylinders get too cold as the mixture will separate out (via lamination) and you can risk giving pure, hypoxic, nitrous to your patient.
@anaestheasier ♬ original sound - Anaestheasier
So should I use it?
Yes. Or no. Your choice.
I realise that's not overly helpful.
Personally I only use it for two occasions:
- When doing a general anaesthetic for a category 1 c-section, to reduce how much sevoflurane and opioid analgesia I have to give
- When doing a general anaesthetic for drainage of an abscess - this peculiar operation has a single moment of intense pain at incision, followed by comparatively little stimulation as they drain and pack the wound. I find nitrous very useful for analgesia and preventing laryngospasm at the point of incision, after which I can switch it off again
The dreaded isotherm
This graph might come up in your FRCA Primary SOE or OSCE, and you need to have something remotely sensible to say about it.
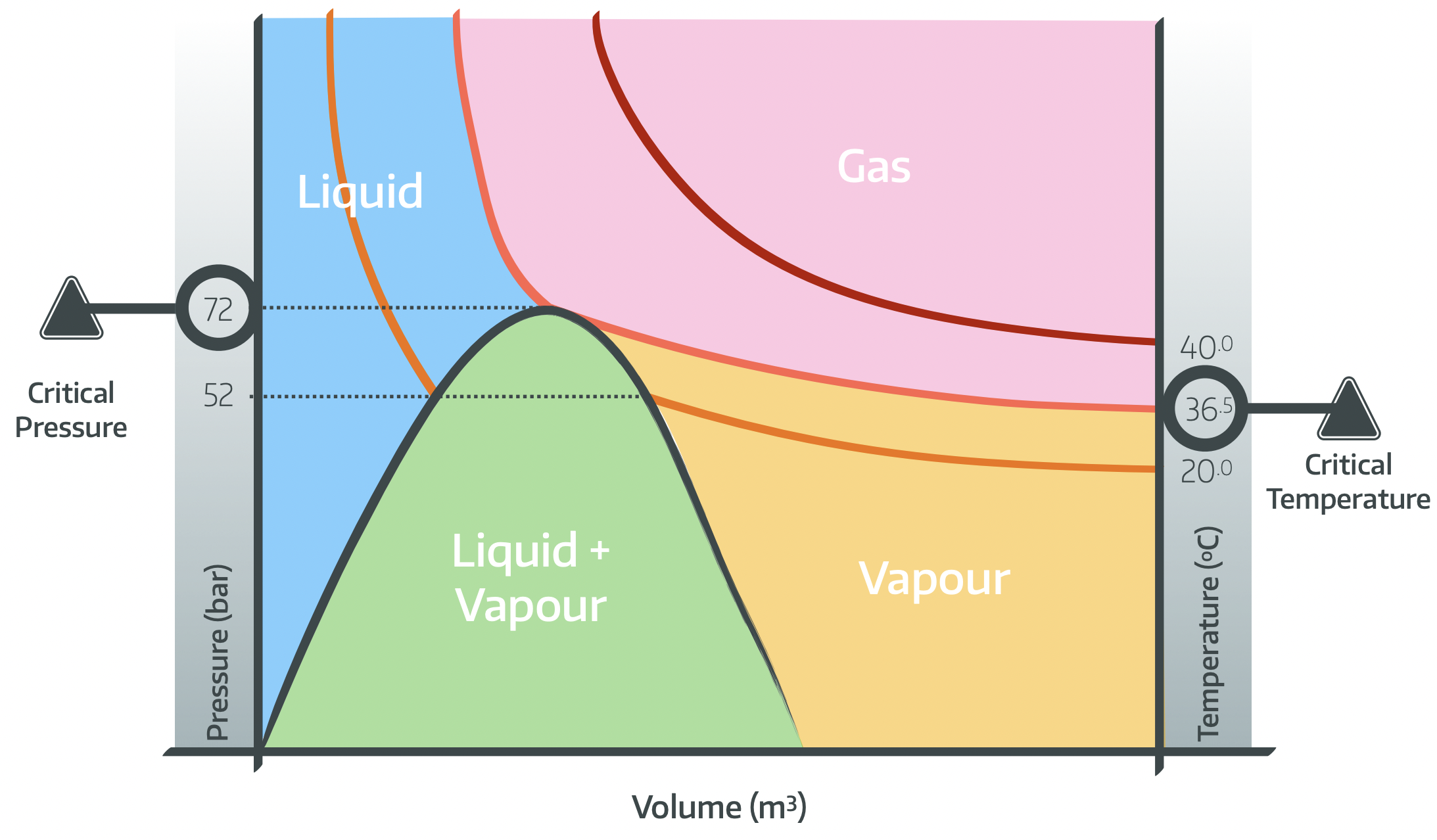
The easiest thing to do is probably just watch our video here:
Useful Tweets
Nitrous oxide pioneer Gardner Quincy Colton was born #OTD in 1814. He demonstrated its anaesthetic use all over America and Europe, leading Joseph Clover to adapt his chloroform apparatus for nitrous oxide. Today's #TriviaTuesday object! 👀👇 #histmed #MedTwitter pic.twitter.com/d0Z6R7pyVO
— Anaesthesia Heritage (@Anaes_Heritage) February 7, 2023
🔓Autonomy vs. the environment: nitrous oxide use in obstetrics, an ongoing challenge.
— 𝘈𝘯𝘢𝘦𝘴𝘵𝘩𝘦𝘴𝘪𝘢 (@Anaes_Journal) January 31, 2023
"The ideal analgesic drug in labour should be easy to administer, affordable, rapidly acting, safe for mother and baby and not hinder the progression of labour."
🔗https://t.co/cPnf5milUa pic.twitter.com/FAngGRXqoL
References and Further Reading

Primary FRCA Toolkit
Members receive 60% discount off the FRCA Primary Toolkit. If you have previously purchased a toolkit at full price, please email anaestheasier@gmail.com for a retrospective discount.

Discount is applied as 6 months free membership - please don't hesitate to email Anaestheasier@gmail.com if you have any questions!
