Dead Space and West Zones
Take home messages:
- Ventilation/Perfusion matching is everything (sort of)
- Dead space is important to think about, especially in paediatrics
Podcast episode
What is V/Q matching?
To survive, we need to get oxygen from the air into our blood, in order to supply the tissues.
This is why you start assessing your patient with an airway, breathing, circulation approach, because that's the most life-threatening order in which problems present.
The lungs represent an interface between ventilation and perfusion. Not only does the air need to be drawn successfully into the alveoli, but blood needs to be pumped to those same alveoli in order to pick up the oxygen.
If the air and the blood are being sent to different alveoli, then clearly the system isn't going to work very well, and this is called a ventilation/perfusion mismatch.
What is dead space?
Dead space refers to the areas of the respiratory tract that are ventilated but not perfused. These areas therefore do not undertake gas exchange with the blood.
The total dead space is called the physiological dead space. It is composed of anatomical and alveolar dead space.
Why does it matter?
Because if you take lots of rapid shallow breaths, you will be moving air in and out of your dead space, without sending any meaningful ventilation to the areas of your lung that can engage in gas exchange.
Likewise if you have an adult patient on a ventilator and they're only achieving tidal volumes of 200ml, it's likely that most of that is dead space ventilation, and they're likely to become steadily more hypoxic and hypercapnoeic.
Physiological dead space = Anatomical dead space + Alveolar dead space
Anatomical dead space
- Anatomical dead space refers to the volume occupied by the conducting airways that supply the alveoli, but don’t undertake gas exchange themselves, and this is generally the first 16 airway generations
Alveolar dead space
- Alveolar dead space refers to the alveoli that are ventilated but do not receive enough blood to undertake gas exchange
- This can be physiological, such as in hypoxic pulmonary vasoconstriction, or pathological, as seen in pulmonary embolism
How can you measure dead space?
Total or physiological dead space is measured using Bohr’s equation.
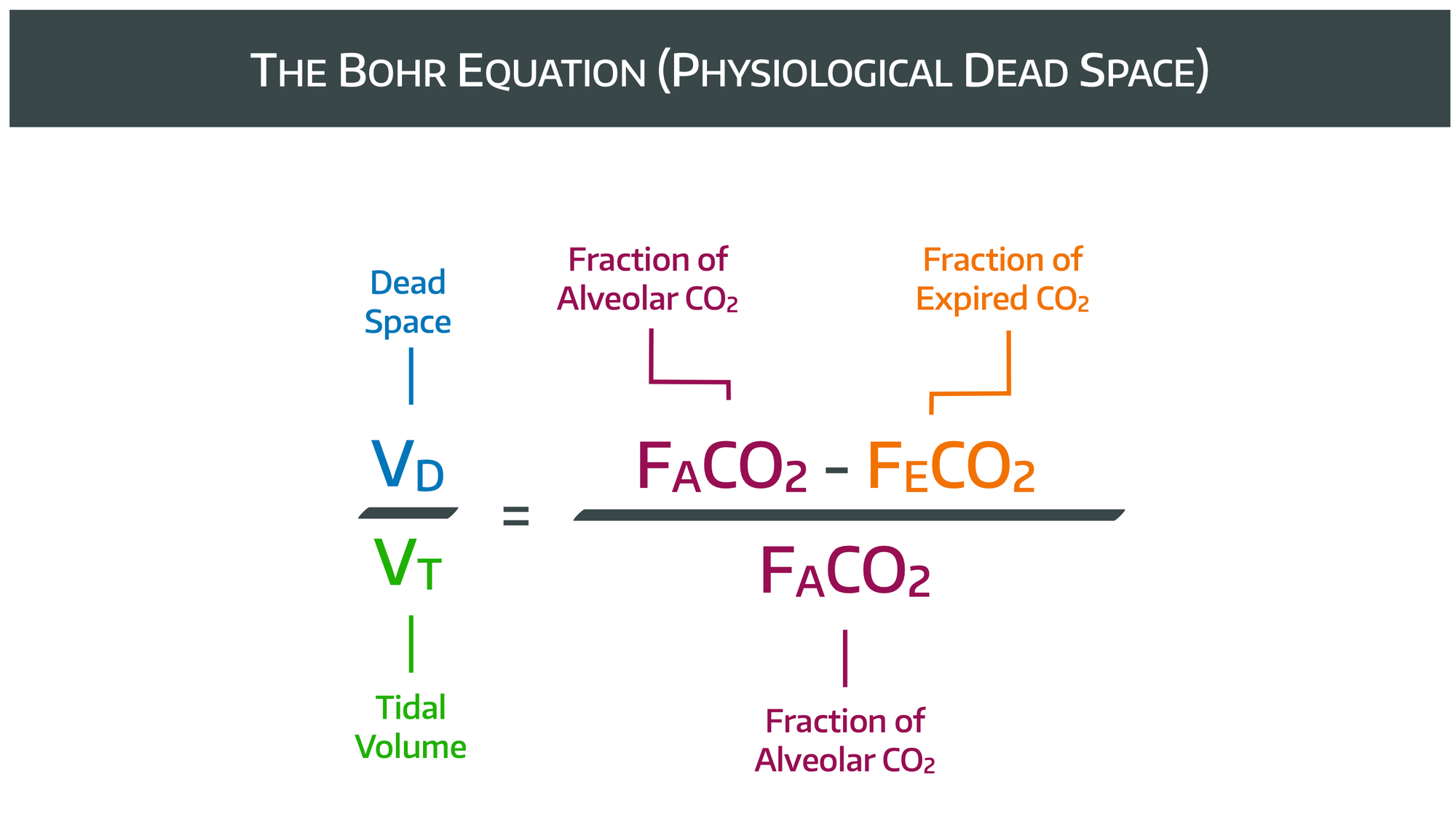
It is usually around 200-350ml in normal tidal breathing.
The explanation
We can reasonably assume that a tidal volume is comprised of alveolar volume and dead space volume, and we can also reasonably assume that inspired CO2 is minimal, if there is no rebreathing occurring.
Therefore the entire expired CO2 is only going to be coming from the alveolar ventilation. Therefore we can generate the equations below, and rearrange as demonstrated to form the Bohr equation.

The Enghoff modification simply extends this further with another assumption.
This assumption is that the alveolar partial pressure of CO2 can reasonably be approximated by the arterial partial pressure of CO2, which makes it all a lot easier to measure.
This works fairly well as long as you remember that this assumption will usually over estimate dead space, as arterial CO2 is slightly higher than alveolar, and will also be affected by:
- Dead space
- Shunt
- Diffusion impairment
Measuring anatomical dead space
Anatomical dead space is measured using Fowler’s method.
It is approximately 2ml/kg.
Step 1
Patient takes a vital capacity breath of pure (100%) oxygen, thereby removing all nitrogen from the anatomical dead space (Remember that the alveoli still receive nitrogen from the blood).
Step 2
Patient exhales all the way to residual volume into a pneumotachograph, which measures flow over time and therefore provides a volume measurement.
(flow/time = volume)
Step 3
The concentration of nitrogen detected is plotted against volume to look like the graph below, where Area A = Area B.
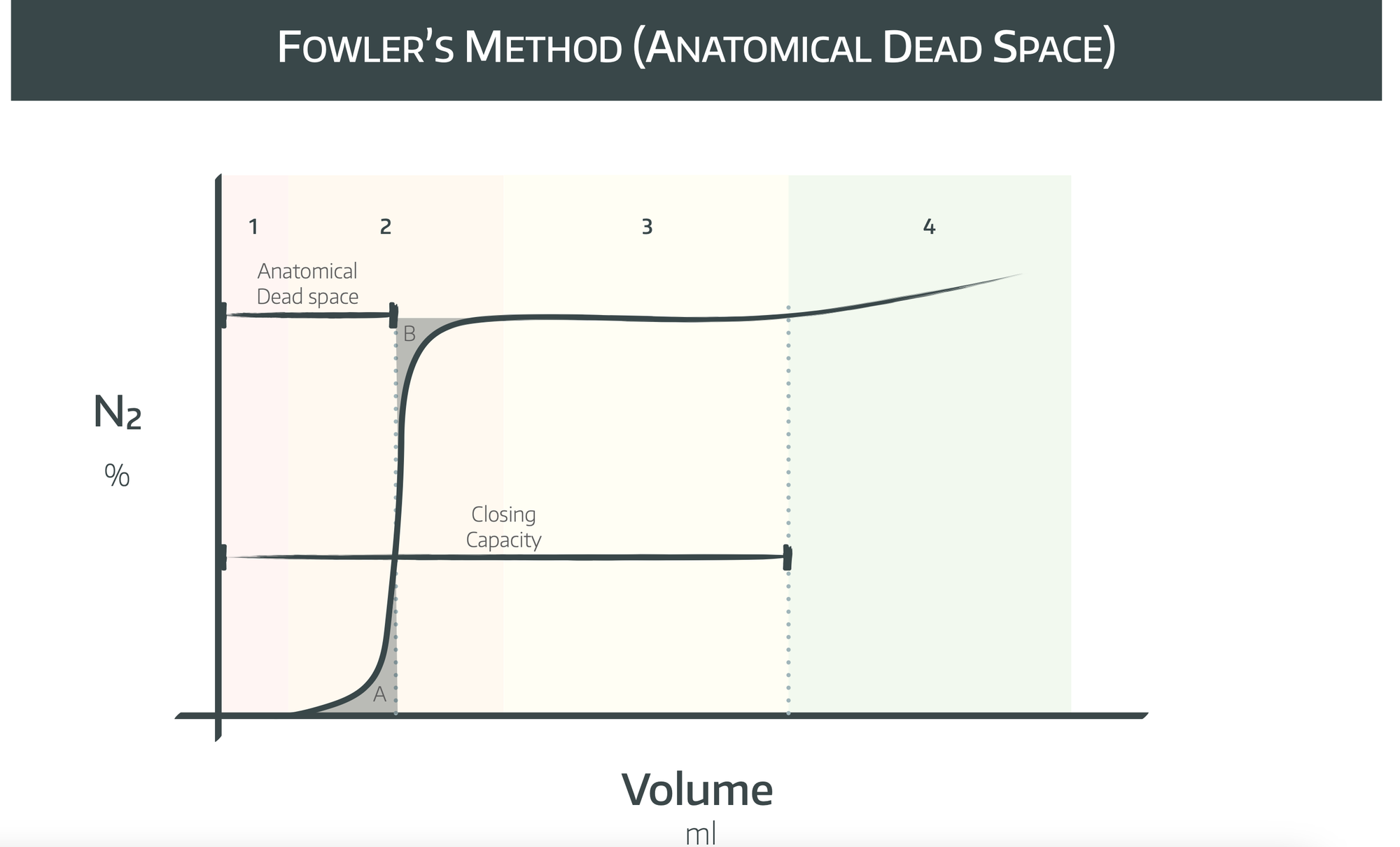
To begin with, pure oxygen is exhaled from the dead space, so no nitrogen is detected - this is phase 1.
In phase 2 you’re detecting a gradually increasing concentration of nitrogen, because different alveoli have different time constants. This means that some alveoli will empty their nitrogen before others, so there will be mixing of alveolar and dead space gas.
Towards the end of this phase, most of the dead space oxygen will have already been exhaled, and more alveoli will be now be draining their nitrogen and therefore the nitrogen concentration detected increases.
In phase 3 there is a plateau, representing only alveolar ventilation, as all dead space gas has now been exhaled.
In phase 4 there is an inflection and a sudden increase in the nitrogen concentration, and this occurs at the closing capacity.
The reason this occurs is because the basal alveoli are more compliant than the apical alveoli, and as a result receive more oxygen during the initial breath of 100% oxygen, so the concentration of nitrogen is actually higher in the apical alveoli, as less of the nitrogen is washed out in that initial breath.
The basal alveoli drain first during the exhalation, until the closing capacity is reached and the apical alveoli are the only ones left to drain. As a result, the nitrogen concentration detected increases suddenly on the graph.

Step 4 (well done keep going!)
Now draw a vertical line approximately half way through phase 2, such that the highlighted areas A and B are equal. This line cuts through the x axis at the anatomical dead space volume
Fowler originally guesstimated this to be a reasonably sensible cut off point, and then tested it with a bunch of rubber models to show that it worked. There is no algebraic explanation that I can find as to exactly why this is the point at which you draw the vertical line.
What about alveolar dead space?
This is really hard to measure.
Fortunately, we've calculated total dead space using Bohr's equation, and anatomical dead space using Fowler's method, so we can just use the equation:
Total = Anatomical + Alveolar
To subtract anatomical from total to tell us our estimated alveolar dead space.
Simples.
What are West zones?
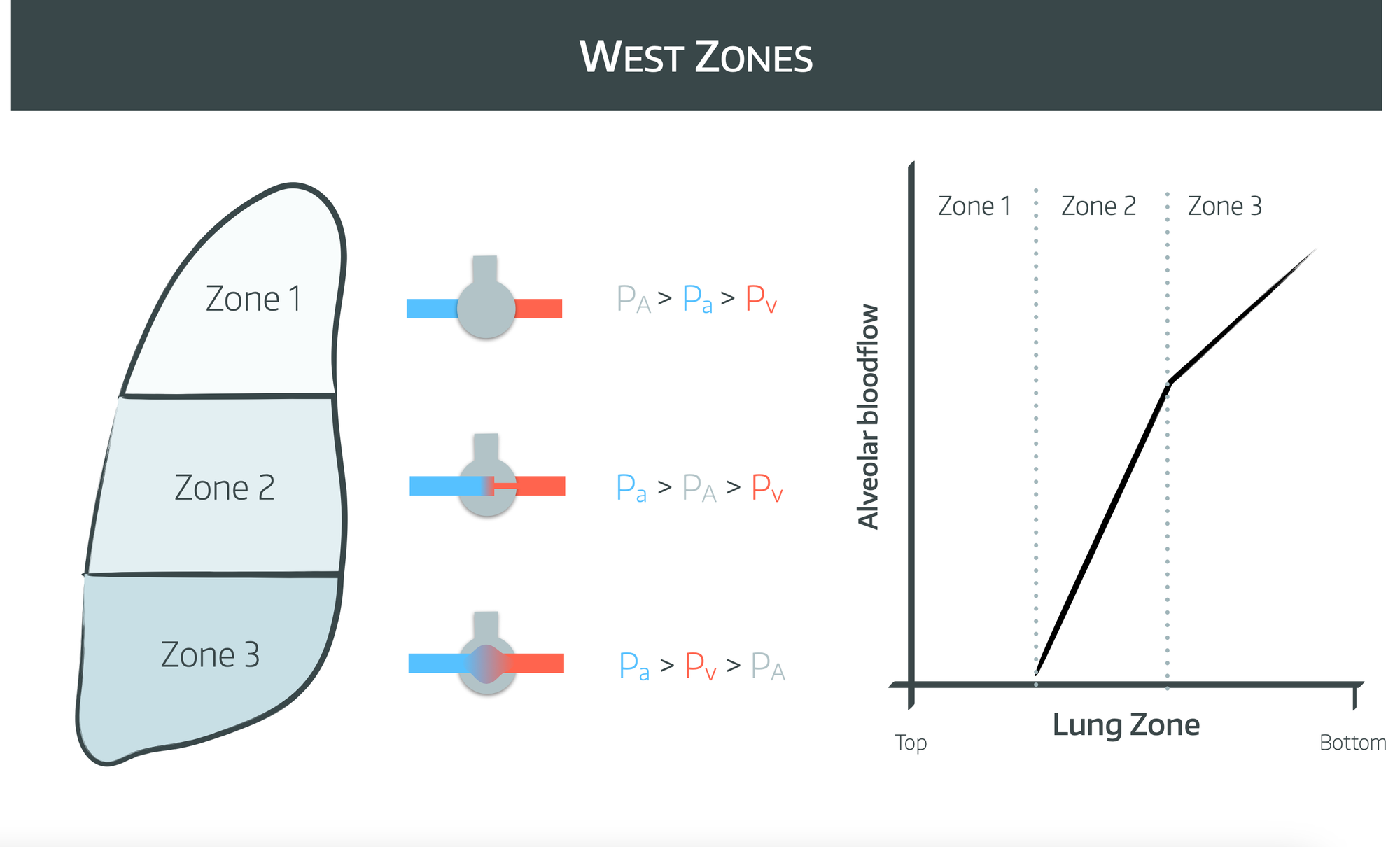
When in the upright position, blood distribution to the different areas is divided into zones called ‘West Zones’. Classically there are three zones described, but a fourth has been added to account for low lung volumes.
Zone 1
- PA>Pa>Pv
- Ventilation is far higher than perfusion as arterial and venous blood vessels are compressed by the higher intra-alveolar pressure
- This zone is generally only really present during positive pressure ventilation, where alveolar pressures are far higher than during negative pressure breathing
- Tendency to form dead space
Zone 2
- Pa>PA>Pv
- Perfusion depends on the difference between arterial and alveolar pressures and varies with both cardiac and respiratory cycles
- Perfusion is higher at the bottom of zone 2 than at the top
Zone 3
- Pa>Pv>PA
- Given both arterial and venous pressures are higher than alveolar pressure, blood flow is consistent to this zone
- It represents areas of shunt
Zone 4
- Very similar to zone 3 but with higher resistance
What is normal V/Q matching?
- V represents ventilation, which is usually around 4 - 5 litres per minute
- Q represents perfusion, which is usually around 5 litres per minute
- A normal V/Q ratio is therefore around 0.9
How does V/Q matching vary with zones of the lung?
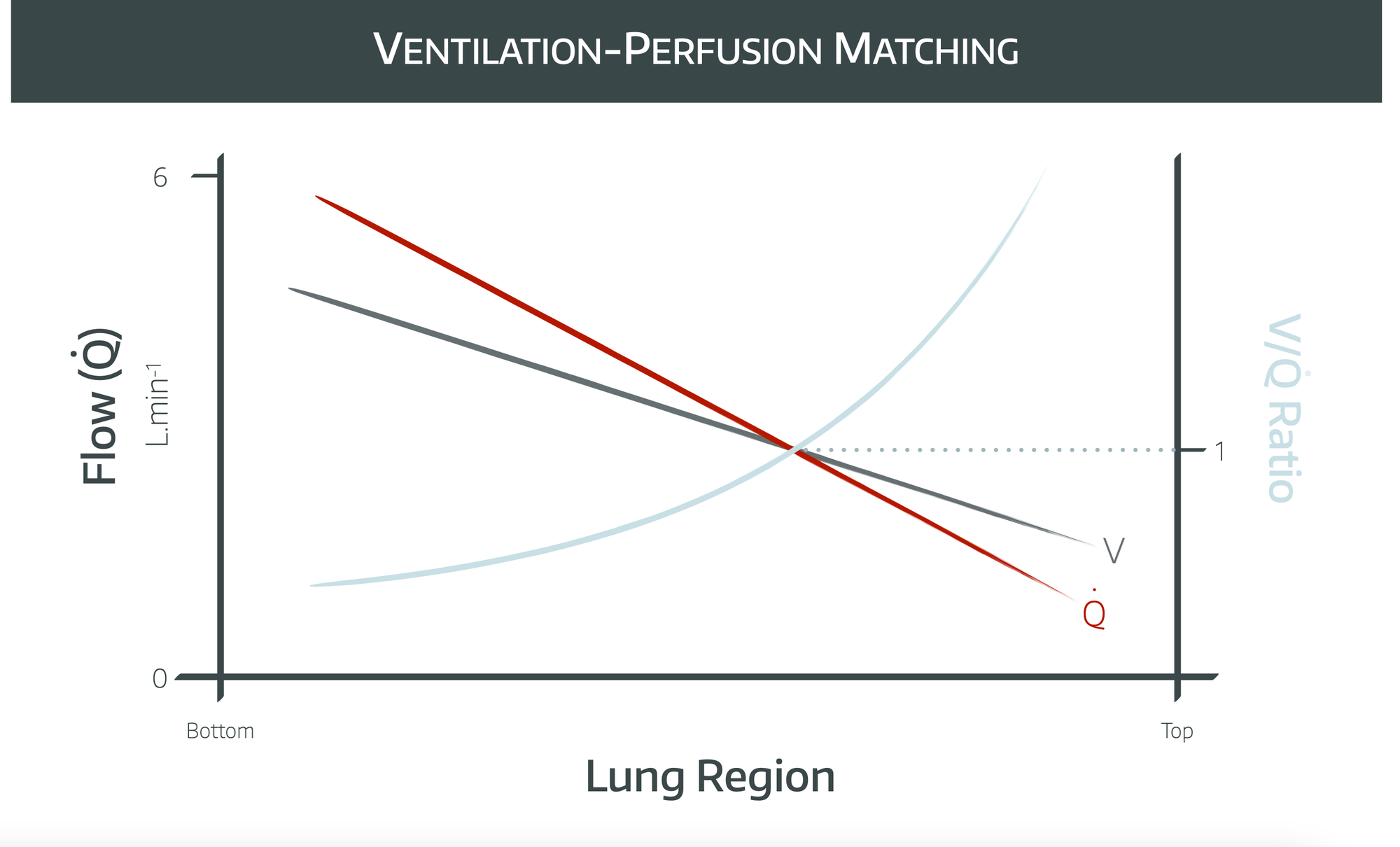
- Both perfusion and ventilation gradually decrease as you travel from the bottom to the top of the lung, but perfusion decreases to a greater extent
- This means that VQ ratio is lower at the bottom and increases towards the top of the lungs
- Therefore the bottom of the lungs demonstrates shunt and the top demonstrates dead space