Local Anaesthetics

Take home messages
- Local anaesthetic agents block voltage gated sodium channels
- They will do this in the heart and brain if you let them
- pKa determines speed of onset
- Fat solubility determines potency
- Duration of action is largely determined by protein binding
Podcast Episode
A spot of history
If you ignore the heart attacks, addiction and illegality, cocaine is actually pretty awesome. The Ancient Peruvians would chew the leaves or mix it with lime and ash to release the active alkaloid, and they used it extensively for a number of purposes including but not limited to:
- Suppressing hunger
- Increasing cold tolerance
- Improving endurance
- Spiritual rituals
- Analgesia for a wide variety of surgical procedures including trephining, cauterising and even bone transplants..?
While drooling cocaine-laced saliva into an open head wound doesn't sound like optimal 2023 management, it did a very good job of numbing the scalp at a time when ultrasound was somewhat harder to come by.
In 1860 Albert Niemann isolated cocaine in crystalline form and in 1884 Karl Koller and Joseph Gärtner dropped cocaine solution into a frog’s eye, noting that it abolished the corneal reflex. They then tried on each other and stuck pins in their eyes for fun.
1908 Bier uses procaine for ‘direct vein anaesthesia’, now known as Bier’s block.
There are two different types
The first thing to blurt out in your FRCA Primary SOE question about local anaesthetic agents is that there are two types:
- Amides - lidocaine, levobupivacaine, prilocaine
- Esters - cocaine, procaine, tetracaine
They all share the same basic structure:
- An aromatic group with a benzene ring at one end
- An amine group with a nitrogen at the other end
- A linkage group in the middle - either an ester or an amide, hence the names
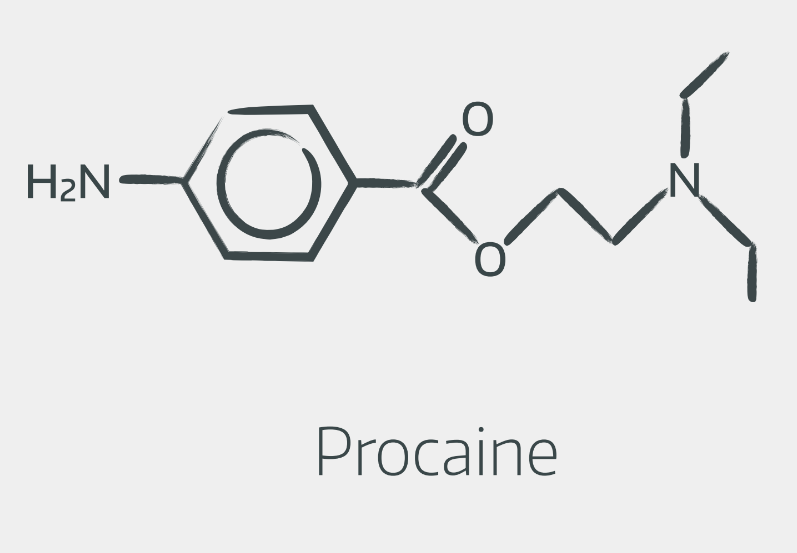
Esters look a bit like this
Amides look a bit like this
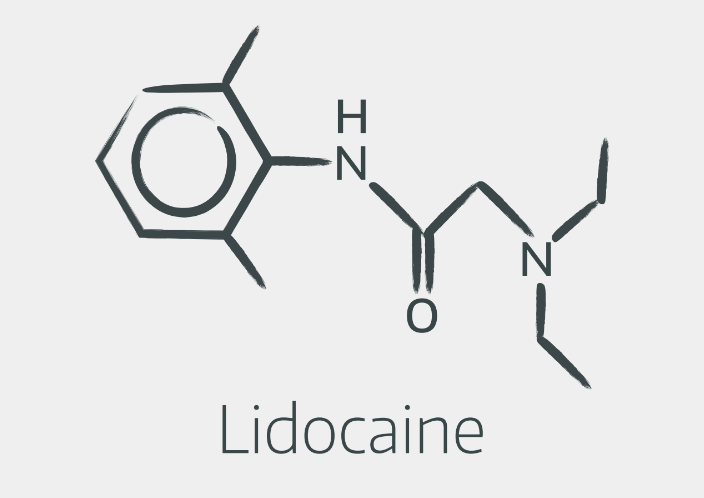
You don't need to be able to draw them out, however it's helpful to recognise the structures, and understand that the linkage in the middle is responsible for the two groups' different metabolism.
Generally speaking we tend to stick to the amides in anaesthesia because they're faster onset and cause fewer allergic reactions, but the ENT surgeons still enjoy a splash of cocaine now and again, and if you use ametop cream then you're using an ester.
How they work
Here's your headline answer for the viva question:
"Local anaesthetic agents induce reversible, use-dependent blockade of voltage-gated sodium channels."
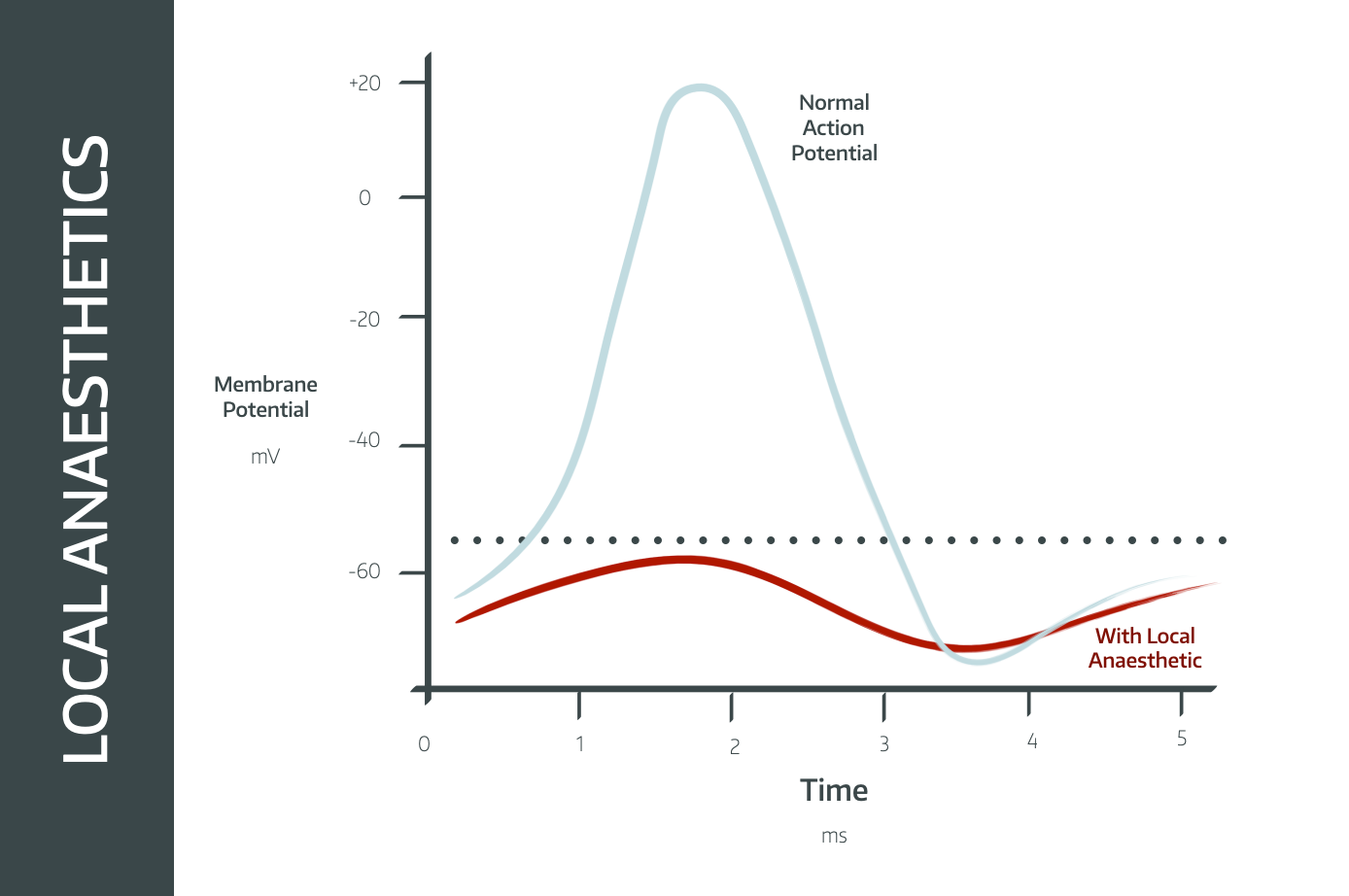
They act on the intracellular aspect of the channel, affecting the smallest nerves first, and larger nerves more slowly.
- This is why the different functions are affected in the following order:
- Dull pain first (smallest C-fibres)
- Temperature and sharp pain
- Light touch
- Pressure
- Motor neurones last (largest Alpha fibres)
Other bits you don't need to know
They also bind to a lesser extent to:
- potassium channels
- calcium channels
- G-protein coupled receptors
which may play a role in dextrobupivacaine’s cardiotoxicity.
'Membrane Expansion' is another possible theory:
- By infiltrating the phospholipid bilayer and altering the interface between the sodium channel and the membrane itself, it may render the channel inactive
For the exam, however, focus on the sodium channel mechanism.
They have different properties
Rather than using the same drug every time, we can take advantage of the fact that local anesthetic agents each have different properties which make them really useful for different things.
- Lidocaine starts working very quickly but it doesn't last very long so it's perfect for little skin-based procedures - such as cannulation or suturing - where you need it to be numb quickly but not for very long
- Bupivacaine takes a little bit longer to work but it lasts much longer - perfect for spinal anaesthesia for hip and knee replacements that take around 90 minutes
- Prilocaine is very fast acting and lasts maximum 60-90 minutes, making it perfect for smaller day case procedures
Safe Doses
You need to be able to spout off the correct maximum safe doses of local anaesthetic agents, even though there's not much evidence for them and they're best guesses based on animal data, meaning we're probably underdosing our patients rather than giving too much.
Giving adrenaline induces vasoconstriction, reducing how much anaesthetic is absorbed sytemically and therefore allowing you to give a little more, depending on the drug.
What are the maximum safe doses of amide local anaesthetic agents, with and without adrenaline?
- Prilocaine - 6mg/kg (9mg/kg with adrenaline)
- Lidocaine - 3mg/kg (7mg/kg)
- Ropivacaine - 3.5mg/kg (3.5mg/kg)
- Bupivacaine - 2mg/kg (2.5mg/kg)
- Levobupivacaine - 2.5mg/kg (3.0mg/kg)
Bupivacaine is hardly affected by adrenaline because it's so protein bound. Lidocaine and prilocaine are much less protein bound, and so there is a real benefit to inducing vasoconstriction.
I remember lidocaine's doses using syllables:
- Li-do-caine = 3 syllables = 3mg/kg
- Li-do-caine-ad-ren-a-line = 7 syllables = 7mg/kg
A bit about pKa
The idea of pKa is weird, but if you understand it then it explains why the different local anaesthetics behave in the way that they do.
- Most drugs are either basic or acidic in nature
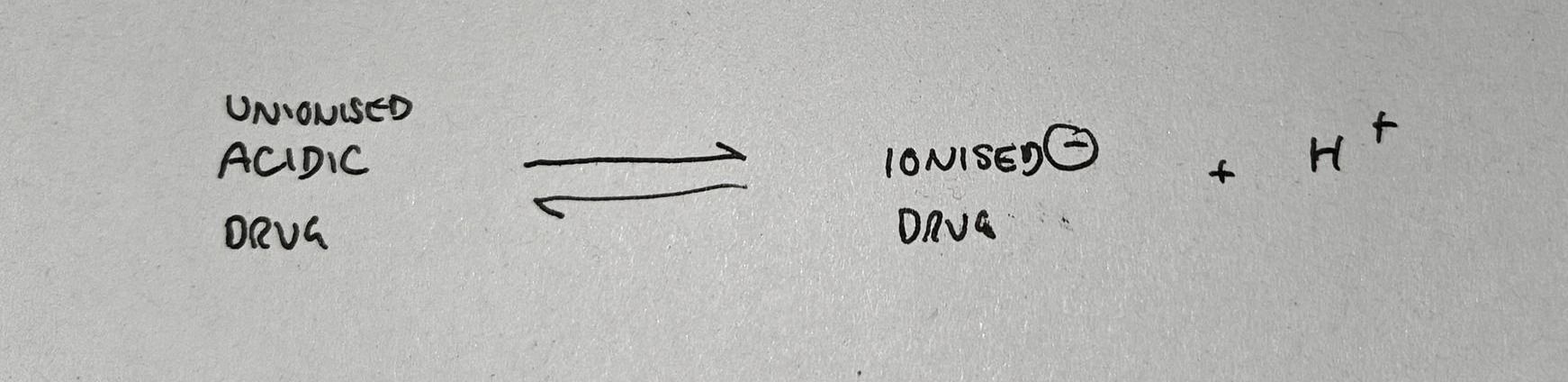
- Acidic drugs can ionise by giving up a proton (H+) and becoming negatively charged
- Basic drugs can ionise by accepting a proton and becoming positively charged
If you put an acidic drug into an alkaline environment, it will ionise and give up a proton, because this equation is dragged to the right by the lack of H+ ions in the environment.
Likewise if we take a basic drug and put it into an acidic environment, it will ionise to mop up these extra H+ ions.
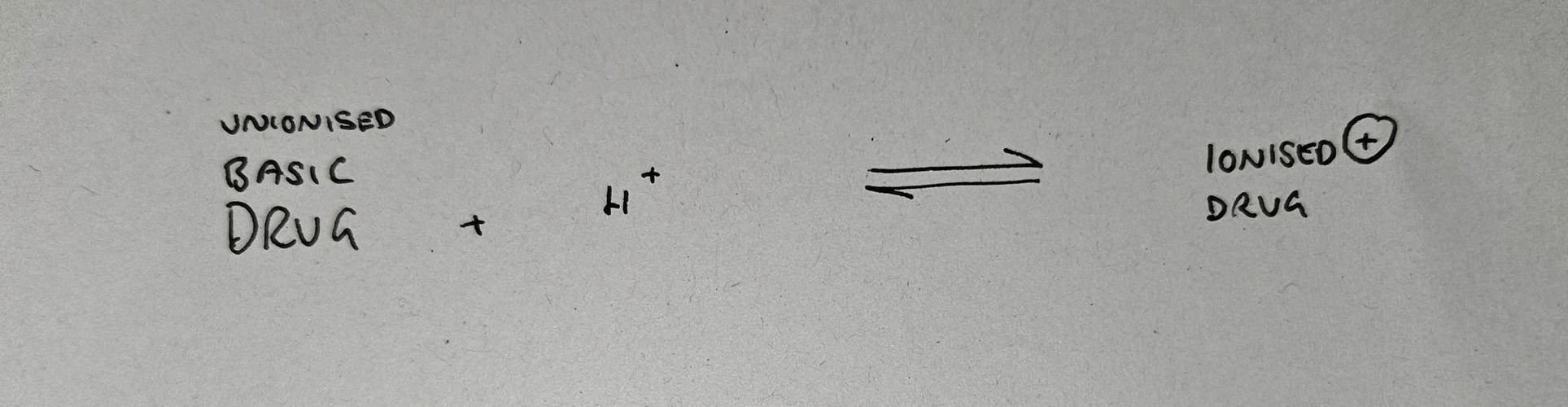
The pKa is simply the pH value at which 50% of the drug is ionised, and 50% is unionised.
- Each drug has its own pKa value
- An acidic drug will be mostly ionised at a pH above its pKa
- A basic drug will be mostly ionised at a pH below its pKa
Why this is even remotely relevant
- Lidocaine and bupivacaine are both basic drugs
- Lidocaine has a pKa of 7.9
- Bupivacaine has a pKa of 8.1
- At a pH of 7.4, lidocaine is 75% ionised, wherease bupivacaine is over 80% ionised
- The local anaesthetic molecule has to get into the cell to bind to the intracellular aspect of the sodium channel in order to work
- Only the unionised fraction can cross the cell membrane
- So 25% of the lidocaine can cross the membrane, but less than 20% of the bupivacaine can
- This is why lidocaine is faster onset than bupivacaine
You see a similar thing with fentanyl (pKa 8) and alfentanil (pKa 6.5)
Here's pKa explained in 60 seconds
Pharmacokinetics
As always it's best to structure your answer to any sort of pharmacokinetics question as follows:
Absorption
- Rapid first pass metabolism if given orally with very low bioavailability
- Usually given subcutaneously, epidurally or IV
- The rate of systemic absorption depends also on the site of injection. From highest systemic absorption to lowest, the order is:
- Intercostal
- Caudal
- Epidural
- Brachial Plexus
- Subcutaneous
Distribution
Protein binding is key to distribution, as a more protein bound drug has much less freedom to move through the tissues and therefore takes longer to be washed away by the blood.
- Bupivacaine >95%
- Ropivocaine 94%
- Lidocaine 70%
- Prilocaine 50%
Lidocaine is approximately 70% protein bound, mostly to albumin and alpha-1-acid glycoprotein
- alpha-1-acid glycoprotein has greater affinity but albumin is far more abundant in the blood so accounts for a greater proportion of lidocaine’s protein binding
- Esters are significantly less protein bound than the amides
Metabolism
Esters generally undergo hydrolysis in the plasma, apart from cocaine which does so in the liver
- May produce PABA (para-aminobenzoate) as a biproduct
Amides undergo slower hepatic metabolism
- 90% of lidocaine undergoes hepatic metabolism by CYP 3A4 and CYP 1A2
- It has an active metabolite MEGX (monoethylglycinexylide)
Excretion
- Renal excretion
Lidocaine vs Bupivacaine
They sometimes ask you to compare lidocaine and bupivacaine, so we made a video.
Do you know any topical local anaesthetics?
Yes. Yes you do.
EMLA
Eutectic mixture of local anaesthetics
- Lidocaine 2.5mg/ml and
- Prilocaine 2.5mg/ml
Eutectic means a mixture of substances in a fixed proportion that melts at a lower temperature than iether of the two constituents.
Note that prilocaine’s metabolite o-toluidine can trigger methaemoglobinaemia in susceptible individuals - this gets asked at the final FRCA and it's usually a child that goes grey and cyanotic despite adequate arterial blood gases.
Amethocaine
Ester local anaesthetic
- 0.5-1% eye drops
- Ametop 4% cream
Causes histamine release which can produce a useful vasodilatation for cannulation and is faster onset than EMLA.
Safe to use in methaemoglobinaemia.
Additives
Like slapping a twin supercharger on a Vauxhall Astra, we can add extra bits and bobs to our local anaesthetic to spice things up a bit.
Adrenaline
Induces vascoconstriction and therefore increases:
- Duration of action
- Potency
- Safe dose for some agents
All three of these are achieved by reducing systemic spread of the local anaesthetic agent.
Opioids
- Synergistic increase in analgesia
- Increased duration of block
- Often will increase speed of onset as well (diamorphine in spinal or fentanyl in epidural)
Steroids
- Increased duration of action and analgesic effect
Clonidine
- Increased analgesic effect
- Can be sedating
Bicarbonate
- Increases speed of onset by increasing unionised fraction of local anaesthetic
Side Effects and Toxicity
As you might imagine, a drug that blocks sodium channels wherever it happens to be can start to cause trouble when found in sufficient doses in the wrong place

Brain
The brain has a high lipid content so local anaesthetics diffuse in rapidly and have a two-phase effect.
Firstly the inhibitory interneurones are blocked, causing excitation
- This manifests as tingling around the mouth
- visual disturbance
- dizziness and tremor
- seizures
Then all of the neuron types are inhibited, leading to coma and finally apnoea
Heart
Unsurprisingly any drug that messes with voltage gated sodium channels is likely to wreak havoc on the cardiac action potential. This can be useful in some circumstances, hence lidocaine is used as an antiarrhythmic agent in certain situations, however there is substantial risk of toxicity with the other local anaesthetic agents, particularly dextrobupivacaine.
- Labile blood pressures
- Tachyarrhythmias
- Ventricular fibrillation
How would you manage local anaesthetic toxicity?
The key here is combinding basic supportive resuscitation with the administration of intralipid, which is literally a big bolus of intravenous fat, to try and coax the local anaesthetic molecules out of the heart and brain.
- Recognition of problem and call for help
- ABC assessement and treatment of reversible issues
- Stop administration of local anaesthetic where possible
- If cardiac arrest, use mechanical CPR
- Intralipid 20%
- 1.5ml/kg over 1 minute as a bolus
- Start Infusion at 15 ml/kg/hour
- After five minutes
- Up to 2 repeat bolus doses 5 mins apart
- Can increase infusion rate to 30ml/kg/hour if cardiovascularly unstable
- not exceeding 12ml/kg total dose
Here's our local anaesthetics summary sheet
Useful Tweets and Resources
Postoperative sore throat? Try alkalinised lidocaine to fill tube cuffs https://t.co/4WSzWTQNvR
— 𝘈𝘯𝘢𝘦𝘴𝘵𝘩𝘦𝘴𝘪𝘢 (@Anaes_Journal) March 29, 2017
References and Further Reading


Primary FRCA Toolkit
Everything you need for the exams in one place.

Discount is applied as 6 months free membership - please don't hesitate to email Anaestheasier@gmail.com if you have any questions!
Just a quick reminder that all information posted on Anaestheasier.com is for educational purposes only, and it does not constitute medical or clinical advice.